 
Lewis structures provide a foundation for understanding the chemical bonding and properties of various compounds. They are denoted by double-headed arrows between the different resonance forms. These structures show different arrangements of atoms while maintaining the same overall connectivity. In more complex molecules, resonance structures may be required to represent the delocalization of electrons. These structures are essential for studying molecular models, predicting molecular shape using the VSEPR theory, understanding atomic orbitals’ hybridization, and determining whether a molecule is polar or nonpolar based on its bond angles. Understanding Lewis structures and their components, such as valence electrons, bonding electrons, and nonbonding electrons, allows us to visualize the electron configuration and chemical structure of different molecules. The Lewis dot structure of PF4 is as follows: The remaining five electrons are placed as l one pairs on the phosphorus atom. To distribute the electrons, we place one electron pair ( two electrons) between phosphorus and each fluorine atom, resulting in four bonding electrons. 
Therefore, the total number of valence electrons in PF4 is 5 + 4 = 9. Each fluorine atom contributes one valence electron. Phosphorus is the central atom, and it has five valence electrons. Let’s take an example of phosphorus tetrafluoride (PF4) molecule. To find the number of nonbonding electrons, we subtract the number of bonding electrons from the total number of valence electrons.īy following these steps, we can construct the Lewis dot structure for various molecules. These electrons are represented by dots in a Lewis structure. Nonbonding electrons, also known as l one pairs, are the electrons that are not involved in bonding and are localized on a single atom. To find the number of bonding electrons in a molecule, we subtract the number of nonbonding electrons from the total number of valence electrons. In a Lewis structure, bonding electrons are represented by lines or dashes between the atoms. Finding Bonding Electronsīonding electrons are the electrons involved in the formation of covalent bonds between atoms. This method allows us to quickly determine the number of valence electrons for most elements. Elements in Groups 13 to 18 have valence electrons equal to their group number minus ten. For example, elements in Group 1 have one valence electron, while elements in Group 2 have two valence electrons. To determine the valence shell electrons, we look at the group number of the element in the periodic table. We place one electron pair (represented by a dot) around each atom until all the valence electrons are used. The central atom is usually the least electronegative element in the compound. Next, we distribute the valence electrons around the atoms in the molecule, starting with the central atom. 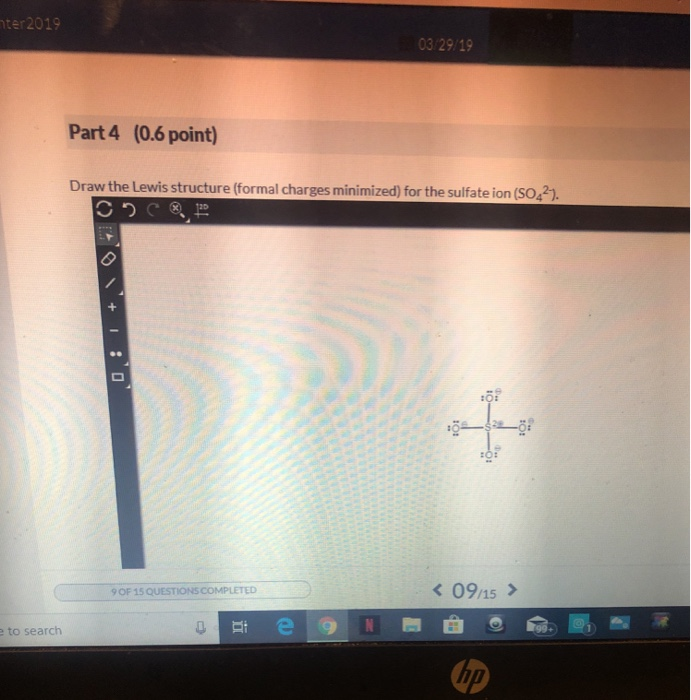
The number of valence electrons can be determined by referring to the periodic table. Valence electrons are the electrons in the outermost shell of an atom and are crucial for chemical bonding. First, we determine the total number of valence electrons in the molecule. To find the Lewis dot structure of a molecule, we need to follow a few steps. By understanding Lewis structures, we can determine the molecular geometry, identify covalent bonds, and predict the overall shape of a molecule. They provide valuable insights into the arrangement of atoms and the distribution of valence electrons. Lewis structures are a visual representation of the chemical bonding and electron pairs in a molecule. 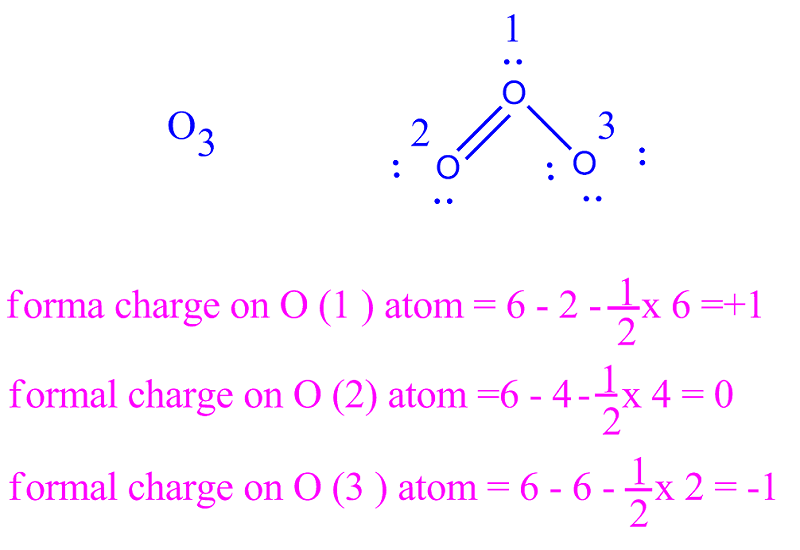
Lewis Structure Molecular Shape PF4 Trigonal Bipyramidal Understanding Lewis Structures 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
